The following is a step by step list on the process of FDA Registration – of companies who manufacture, pack or distribute products under the jurisdiction of the U.S. Food and Drug Administration (FDA) (i.e. food, cosmetics, medical devices or pharmaceuticals)
Registration of Food Exporters in Pakistan (Himalayan Salt) by FDA
Importantly, in case you are a Pakistani company that exports food products or natural products (such as Himalayan salt, herbs, spices, rice, etc.) to the U.S., you are to register your facility with the U.S Food and Drug Administration (FDA) under the Food Facility Registration Program (Section 415 of the FD&C Act).

Step 1: Find out whether FDA Registration is necessary or not
First, make sure that your product and plant are under the jurisdiction of the FDA. The registration of FDA may be required to: All categories are registered and listed differently.

Step 2: Obtain a DUNS Number
A Dun and Bradstreet (D&B) number issued is the Data Universal Numbering System (DUNS): a 9-digit number. FDA refers to this number to ensure that your company is physically located and identified. D&B provides a free application of a DUNS number on the website.
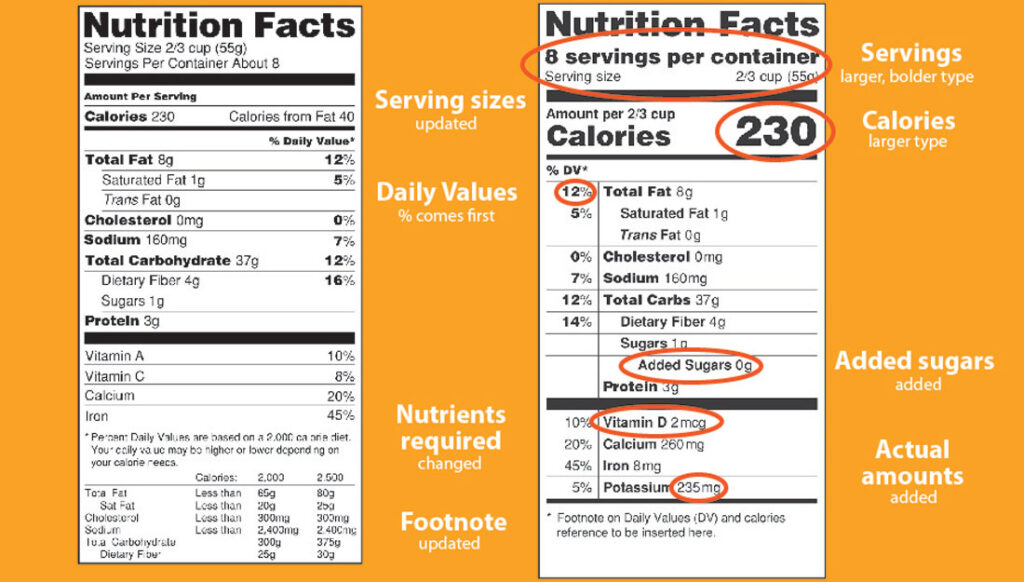
Compliance and Labeling
Make sure that your products are FDA labeled and safe. Good Manufacturing practices (GMP).
Respond promptly to any FDA communications or inspection findings.
FDA Certification: The significance of FDA Certification to the safety and reliability of its products
FDA Certification is a valuable authorization accorded by the U.S. Food and Drug Administration to assure that the products are of high safety, quality and regulation. The certification is applicable in a broad category of products, such as food, medical equipment, cosmetics, drugs, and dietary supplements. A certified product through FDA certification is an item that has undergone the required testing, documentation, and compliance tests that are stipulated by the American law.
In the case of business, FDA Certification develops credibility and consumer confidence. It demonstrates that the company adheres to the right manufacturing design and high-quality standards. In other sectors, including in the pharmaceutical and medical device sectors, product sales in the United States cannot take place without FDA approval. The absence of it can present a business with legal problems, recall, or market bans.
To the consumer, FDA Certification gives the consumer confidence that he is using a safe and reliable product. It guarantees correct labelling, correct ingredients and quality production. It is the health of the people that is safeguarded by FDA; it can be food in the shelf or even a cosmetic product.
On the whole, FDA Certification is essential to protecting and averting risks and enhancing product quality in different industries. It is beneficial to the business as well as the consumer because it enhances trust, transparency, and compliance.

FDA Registration Certificate
Complete the above form and send complete legal document i.e (NTN, SECP,
CHAMBER and any registration or affiliation of the company)
We will apply for DUNS No. (If not available)
For New DUNS registration takes at least 2 weeks
After getting DUNS Registration we directly applied for FFR FDA Registration via
online system.
Each product included in registration required at least 2-3 samples for FDA Activity
Retention
Require In-house Testing Reports of Sampled Products
External Testing Report from any reputed organization i.e PCSIR
The approval of FDA Registration take at least 1 week.
Compliance Networks FDA Registration Process

